|
4/1/2023 0 Comments Vaccine side effect rates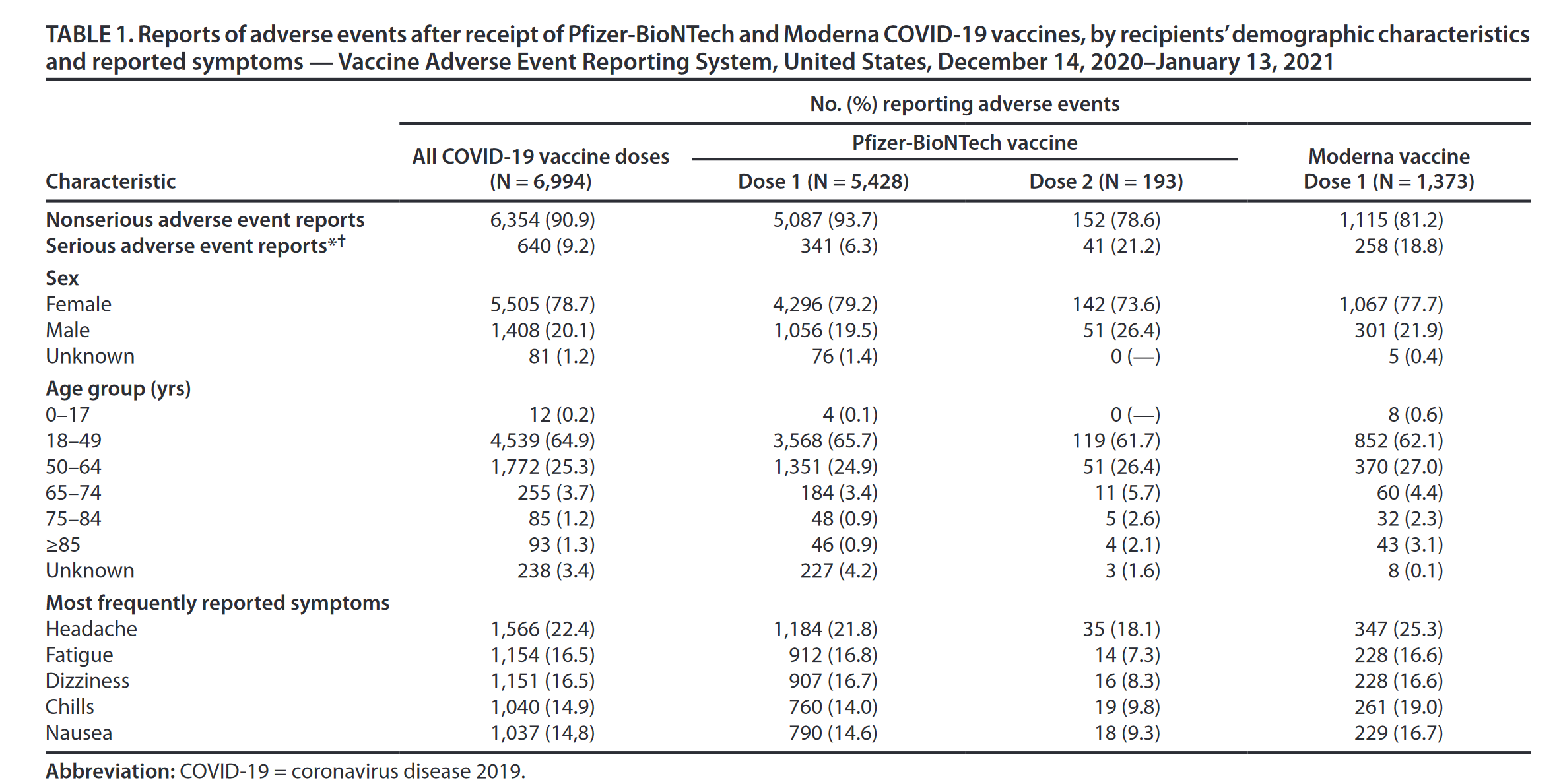
Both showed lower incidence of systemic reactions than non-mRNA counterparts. Both vaccines elicited more frequent and stronger reactions following the second dose of the two-dose series. In the mRNA-1273 trial, axillary swelling/tenderness was reported by 10–14% of individuals. The most common reactions reported in both trials were pain, redness, and swelling at the injection site, fever, headache, fatigue, chills, myalgia, and nausea/vomiting. Trials of both vaccines generated data suggesting high safety and efficacy, with typical and mild local and/or systemic reactions. 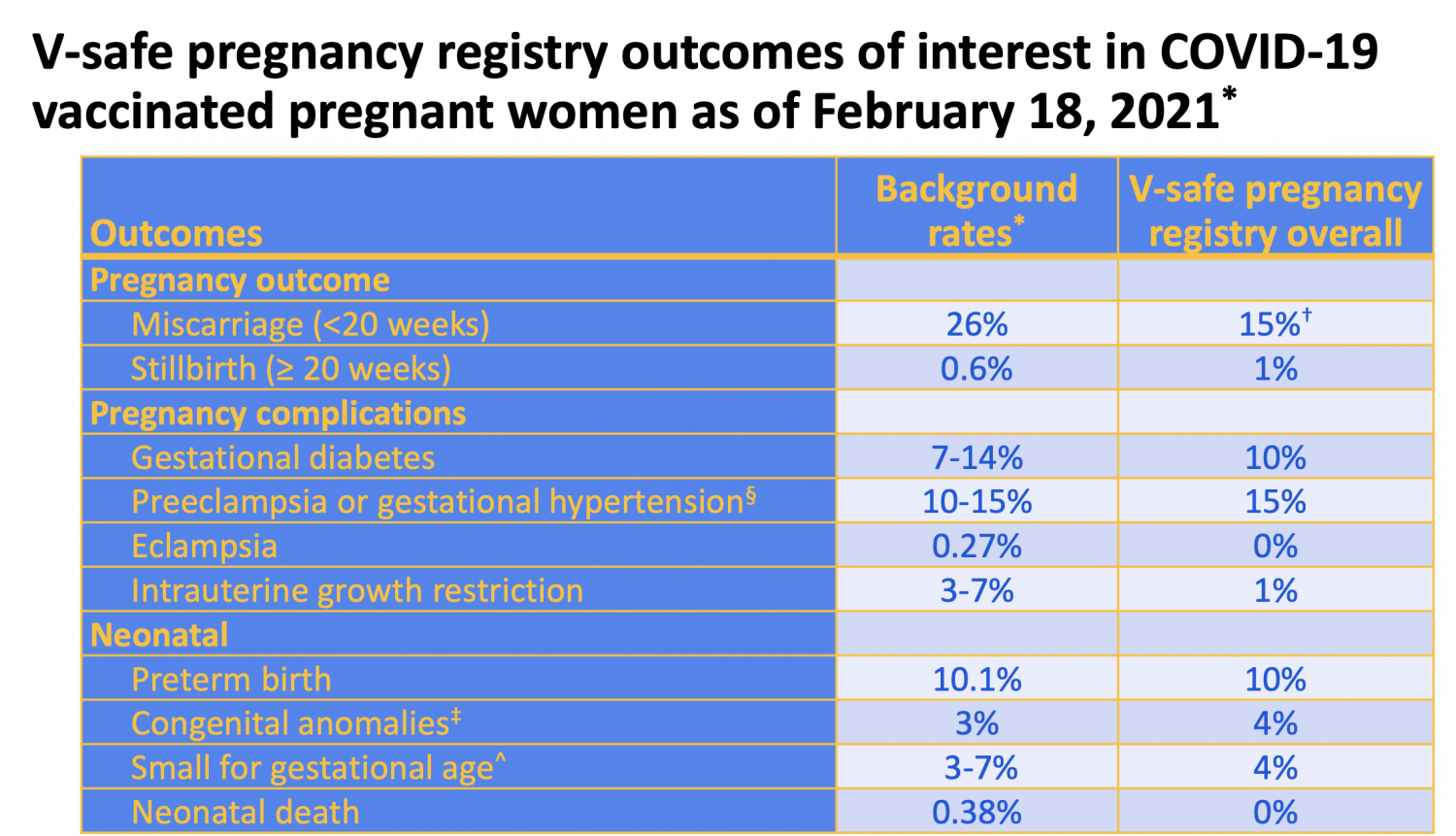
The mRNA-1273 trial utilized a similar design, encompassing 15,163 healthy individuals falling into either the 18 to 64 or 65+ years of age group. Profiles of vaccine reactogenicity to BNT162b2 vaccine were based on an initial sample of 4093 healthy individuals from one of two age groups – 18 to 55 years, and 55+ years of age. citizens on December 14 and 21, respectively.Įmergency Use Authorization was based on data from Phase III clinical trials, including reactogenicity profiles generated from subsets of the clinical data. Following subsequent review and approval of each vaccine by the Centers for Disease Control (CDC), the first vaccinations outside of clinical trials were given to U.S. A second EUA was issued for Moderna’s version of the vaccine (mRNA-1273) one week later, on December 18, 2020. Food and Drug Administration (FDA) approved Emergency Use Authorization (EUA) for the Pfizer-BioNTech mRNA-based COVID-19 vaccine (BNT162b2). Further, we argue this community-based approach could be important in the future in three key ways: 1) as new boosters and modified vaccines re-volatilize vaccine hesitancy, 2) as new vaccines receive similar testing and rapid authorization, and 3) to combat vaccine hesitancy in other arenas (e.g., annual vaccines, childhood vaccines). Our findings allow broad-scale estimates of the nature and severity of reactions one might expect following vaccination within a clinically-diverse community, and provide a context for addressing vaccine hesitancy in communities such as ours, where locally-generated data and communication may be more influential than national trends and statistics in convincing individuals to become vaccinated. The reported severity to the COVID-19 vaccine was positively correlated with self-reported responses to other vaccines. Individuals who reported having previously tested positive for COVID-19 reported stronger responses following the first dose of either vaccine relative to COVID-naïve individuals. Responses to the mRNA-1273 vaccine were more severe than to BNT162b2, though all were generally in the mild to moderate category. In general, the pattern and severity in side effects was similar to both clinical trial data as well as other published studies. Significance was assessed using Bonferroni-adjusted criteria within families of tests. 
The data were analyzed utilizing a range of statistical tests, including chi-square tests of association, Cohen’s h, Kruskal-Wallis test one-way nonparametric ANOVA, least-squares regression, and Wilcoxon signed-ranks test. All statistical analyses were carried out using SYSTAT, version 13. 
Respondents were asked to rate the incidence and severity of 15 potential side effects and two related outcomes following each of their two doses of the vaccine. Individuals that received two doses of either BNT162b2 or mRNA-1273 between Decemand were sent a survey, created by the research team. As new versions of the vaccine are rolled-out, local initiatives such as this may offer a means to combat vaccine hesitancy in reference to COVID-19, but are also important as we face new viral threats that will necessitate a rapid vaccine rollout, and to combat a growing public distrust of vaccines in general. Here we report the results of a survey of 4825 individuals from southcentral Kentucky who received two doses of either the Pfizer-BioNTech (BNT162b2) or Moderna (mRNA-1273) vaccine between Decemand May 1, 2021. The rapid authorization and widespread rollout of COVID-19 vaccines in the United States demonstrated a need for additional data on vaccine side effects, both to provide insight into the range and severity of side effects that might be expected in medically-diverse populations as well as to inform decision-making and combat vaccine hesitancy going forward.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
